What Is DL-6 8-Thioctic Acid?
DL-6 8-Thioctic acid, also known as lipoic acid or alpha-lipoic acid (ALA), is a naturally occurring organosulfur compound that plays a crucial role in cellular energy production and antioxidant defense. This powerful molecule has garnered significant attention in the scientific community due to its unique properties and potential therapeutic applications.
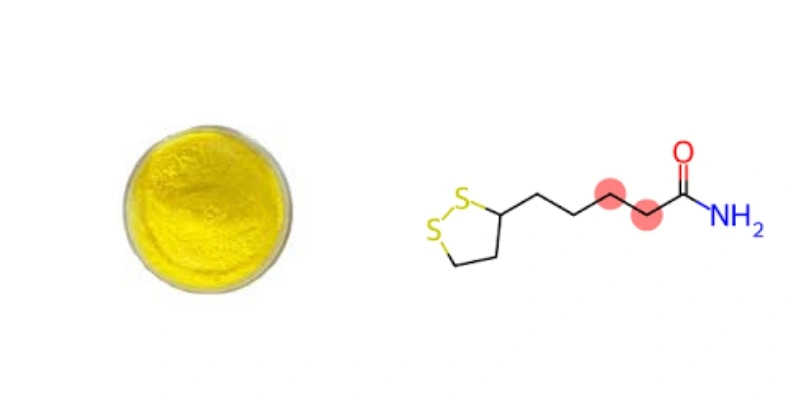
What is the chemical structure of DL-6 8-thioctic acid?
DL-6 8-Thioctic acid has the molecular formula C8H14O2S2 and is characterized by a unique cyclic disulfide structure. The molecule consists of a five-membered dithiolane ring with a carboxylic acid group attached to one of the carbon atoms. The "DL" prefix indicates that it exists as a racemic mixture of two enantiomers: the R-enantiomer (natural form) and the S-enantiomer.
The chemical structure of DL-6 8-thioctic acid is as follows:
- A five-membered dithiolane ring (containing two sulfur atoms)
- A carboxylic acid group (-COOH) attached to the ring
- A four-carbon aliphatic tail
This unique structure contributes to the compound's ability to exist in both oxidized and reduced forms, which is crucial for its biological functions. The oxidized form contains a disulfide bond within the dithiolane ring, while the reduced form (dihydrolipoic acid) has two free thiol groups.
The presence of both hydrophilic and hydrophobic regions in the molecule allows it to interact with various cellular components, including proteins and lipid membranes. This amphipathic nature is essential for its diverse biological activities and therapeutic potential.

How is DL-6 8-thioctic acid synthesized?
The synthesis of DL-6 8-thioctic acid can be achieved through various methods, both in laboratory settings and industrial production. Here are some common approaches to synthesizing this compound:
1. Chemical Synthesis:
One of the most widely used methods for synthesizing lipoic acid powder involves a multi-step process starting from octanoic acid or related compounds. The general steps include:
- Bromination of octanoic acid to form 8-bromooctanoic acid
- Conversion of the bromine to a thiol group
- Cyclization to form the dithiolane ring
- Oxidation to create the final disulfide structure
This method allows for large-scale production but often results in a racemic mixture of R and S enantiomers.
2. Enzymatic Synthesis:
Researchers have explored enzymatic approaches to synthesize DL-6 8-thioctic acid using various microorganisms. This method offers the potential for more stereospecific production, potentially yielding higher amounts of the biologically active R-enantiomer. Enzymes such as lipoic acid synthetase play a crucial role in these biosynthetic pathways.
3. Fermentation:
Some studies have investigated the production of DL-6 8-thioctic acid through fermentation processes using genetically engineered microorganisms. This approach aims to develop more sustainable and cost-effective production methods, particularly for large-scale industrial applications.
4. Green Chemistry Approaches:
In recent years, there has been growing interest in developing more environmentally friendly synthesis methods for DL-6 8-thioctic acid. These approaches often involve the use of renewable resources, catalysts, and milder reaction conditions to reduce the environmental impact of production.
The choice of synthesis method depends on various factors, including scale of production, desired purity, stereochemical requirements, and economic considerations. Ongoing research continues to explore novel and improved synthesis techniques to enhance the efficiency and sustainability of DL-6 8-thioctic acid production.

What are the key properties of DL-6 8-thioctic acid?
DL-6 8-Thioctic acid possesses several remarkable properties that contribute to its biological significance and therapeutic potential. Understanding these key properties is essential for appreciating the compound's diverse applications in research and medicine:
1. Antioxidant Activity:
One of the most notable properties is its potent antioxidant activity. It can directly scavenge reactive oxygen species (ROS) and reactive nitrogen species (RNS), helping to protect cells from oxidative stress. Additionally, it can regenerate other antioxidants such as vitamin C, vitamin E, and glutathione, enhancing the overall antioxidant defense system.
2. Metal Chelation:
DL-6 8-Thioctic acid has the ability to chelate metal ions, particularly transition metals like iron and copper. This property is important for its antioxidant effects and potential neuroprotective benefits, as it can help reduce metal-induced oxidative damage.
3. Redox Activity:
The compound can easily transition between its oxidized (disulfide) and reduced (dithiol) forms. This redox activity is crucial for its role in energy metabolism and its ability to modulate cellular redox status.
4. Amphipathic Nature:
It possesses both hydrophilic and hydrophobic regions, allowing it to interact with various cellular components. This property enables it to cross biological membranes and participate in diverse cellular processes.
5. Coenzyme Function:
In its protein-bound form, lipoic acid powder serves as a crucial coenzyme in several multienzyme complexes involved in energy metabolism, such as pyruvate dehydrogenase and α-ketoglutarate dehydrogenase.
6. Neuroprotective Effects:
Studies have shown that DL-6 8-thioctic acid can cross the blood-brain barrier and exert neuroprotective effects. This property has led to investigations into its potential use in treating various neurological disorders.
7. Insulin Sensitizing Activity:
It has been found to enhance insulin sensitivity and glucose uptake in cells. This property has sparked interest in its potential applications for managing diabetes and metabolic disorders.
8. Anti-inflammatory Effects:
Research has demonstrated that lipoic acid powder possesses anti-inflammatory properties, potentially through its ability to modulate NF-κB signaling and reduce oxidative stress.
9. Stability and Bioavailability:
While DL-6 8-thioctic acid is relatively stable in solid form, it can be sensitive to light and heat in solution. Its bioavailability can vary depending on the formulation and route of administration, with some studies suggesting enhanced absorption through specific delivery systems.
10. Enantiomeric Differences:
The R-enantiomer of DL-6 8-thioctic acid is considered the biologically active form and is naturally produced in the body. However, synthetic preparations often contain a racemic mixture of both R and S enantiomers, which may have slightly different biological activities.
These key properties of lipoic acid powder contribute to its diverse biological functions and potential therapeutic applications. From its role in cellular energy production to its antioxidant and neuroprotective effects, this versatile compound continues to be the subject of extensive research in various fields of medicine and biochemistry.

DL-6 8-Thioctic Acid Manufacturer
At Rebecca Bio-Tech, we're committed to supporting your scientific and industrial needs with top-tier products at competitive prices. Whether you're exploring the antioxidant properties of lipoic acid powder or incorporating it into innovative health supplements, we're here to help you achieve your goals.
Ready to elevate your research or product line with high-quality DL-6 8-Thioctic Acid? We invite you to reach out to our team for more information, sample requests, or to discuss your custom requirements. Contact us at information@sxrebecca.com and discover how Rebecca Bio-Tech can be your trusted partner in advancing your work with DL-6 8-Thioctic Acid.
References:
- Packer, L., Witt, E. H., & Tritschler, H. J. (1995). Alpha-lipoic acid as a biological antioxidant. Free Radical Biology and Medicine, 19(2), 227-250.
- Cronan, J. E. (2014). Biotin and lipoic acid: synthesis, attachment, and regulation. EcoSal Plus, 6(1).
- Koufaki, M. (2014). Therapeutic applications of lipoic acid: a patent review (2011-2014). Expert Opinion on Therapeutic Patents, 24(9), 993-1005.
- Biewenga, G. P., Haenen, G. R., & Bast, A. (1997). The pharmacology of the antioxidant lipoic acid. General Pharmacology: The Vascular System, 29(3), 315-331.
- Suh, J. H., Moreau, R., Heath, S. H., & Hagen, T. M. (2005). Dietary supplementation with (R)-α-lipoic acid reverses the age-related accumulation of iron and depletion of antioxidants in the rat cerebral cortex. Redox Report, 10(1), 52-60.
- Packer, L., Cadenas, E., & Davies, K. J. (2001). Free radicals and exercise: an introduction. Free Radical Biology and Medicine, 31(6), 829-830.





_1730079799361.webp)


